RESEARCH/REVIEW ARTICLE
Environmental influences on the at-sea behaviour of a major consumer, Mirounga leonina, in a rapidly changing environment
Trevor McIntyre,1,2 Horst Bornemann,1 P.J. Nico de Bruyn,2 Ryan R. Reisinger,2 Daniel Steinhage,1 Maria E.I. Márquez,3 Marthán N. Bester2
& Joachim Plötz1
1 Alfred-Wegener-Institut Helmholtz-Zentrum für Polar-und Meeresforschung, Postfach 120161, DE-27515, Bremerhaven, Germany
2 Mammal Research Institute, Department of Zoology and Entomology, University of Pretoria, Private Bag X20, Hatfield, 0028, South Africa
3 Instituto Antártico Argentino, Departamento Biologia de Predadores Tope, Balcarce 290 (C1064AAF), Buenos Aires, Argentina
Abstract
Understanding the distribution and foraging ecology of major consumers within pelagic systems, specifically in relation to physical parameters, can be important for the management of bentho-pelagic systems undergoing rapid change associated with global climate change and other anthropogenic disturbances such as fishing (i.e., the Antarctic Peninsula and Scotia Sea). We tracked 11 adult male southern elephant seals (Mirounga leonina), during their five-month post-moult foraging migrations from King George Island (Isla 25 de Mayo), northern Antarctic Peninsula, using tags capable of recording and transmitting behavioural data and in situ temperature and salinity data. Seals foraged mostly within the Weddell–Scotia Confluence, while a few foraged along the western Antarctic Peninsula shelf of the Bellingshausen Sea. Mixed model outputs suggest that the at-sea behaviour of seals was associated with a number of environmental parameters, especially seafloor depth, sea-ice concentrations and the temperature structure of the water column. Seals increased dive bottom times and travelled at slower speeds in shallower areas and areas with increased sea-ice concentrations. Changes in dive depth and durations, as well as relative amount of time spent during the bottom phases of dives, were observed in relation to differences in overall temperature gradient, likely as a response to vertical changes in prey distribution associated with temperature stratification in the water column. Our results illustrate the likely complex influences of bathymetry, hydrography and sea ice on the behaviour of male southern elephant seals in a changing environment and highlight the need for region-specific approaches to studying environmental influences on behaviour.
Keywords
Southern elephant seals; foraging ecology; satellite-relay data loggers; King George Island; Isla 25 de Mayo; at-sea behaviour.
Correspondence
Trevor McIntyre, Mammal Research Institute, Department of Zoology and Entomology, University of Pretoria, Private Bag X20, Hatfield, 0028, South Africa. E-mail: tmcintyre@zoology.up.ac.za
To access the supplementary material for this article, please see the
supplementary files under Article Tools online.
(Published: 26 November 2014)
Polar Research 2014. © 2014 T. McIntyre et al. This is an Open Access article distributed under the terms of the Creative Commons Attribution-Noncommercial 3.0 Unported License (http://creativecommons.org/licenses/by-nc/3.0/), permitting all non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Citation: Polar Research 2014, 33, 23808, http://dx.doi.org/10.3402/polar.v33.23808
The Antarctic Peninsula and Scotia Sea ecosystems are productive areas within the Southern Ocean (Holm-Hansen et al. 2004; Ducklow et al. 2007) and sustain large populations of land-breeding marine birds and mammals. These areas are also undergoing some of the most rapid environmental changes associated with a warming climate and consequent ice-breakup (de la Mare 1997; Mulvaney et al. 2012). The area supports a large biomass of Antarctic krill (Euphausia superba; Atkinson et al. 2009) and an associated, expanding krill fishery (Nicol et al. 2012). Physical environmental changes (e.g., changes in sea ice and water temperatures) and associated changes in krill abundance (Ducklow et al. 2007; Massom & Stammerjohn 2010) have been linked to population changes in some predators of the region, primarily penguins (Trivelpiece et al. 2011; Naveen et al. 2012). The responses of seal populations to such changes are likely to vary, and ice-breeding seals may be negatively affected by reductions in food availability and loss of sea ice (Siniff et al. 2008; Forcada et al. 2012), while some sub-Antarctic species in this area—notably southern elephant seals (Mirounga leonina)—may benefit for instance through the creation of additional breeding habitat (e.g., Gil-Delgado et al. 2013).
Hydrographically, the Antarctic Peninsula and Scotia Sea ecosystems are strongly influenced by interactions of the Antarctic Circumpolar Current (ACC) with bathymetric features constraining its eastward flow (e.g., the Drake Passage [Cunningham et al. 2003]). Such interactions are particularly pronounced in the Scotia Sea where the uneven bottom topography associated with the Scotia Arc interacts with the ACC to promote strong mixing of the water column (Naveira Garabato et al. 2002). The Weddell–Scotia confluence (bounded by the Weddell Front to the south and the southern boundary of the ACC to the north [Whitworth et al. 1994]), in particular, supports substantial seasonal phytoplankton blooms often associated with eddy activity (Kahru et al. 2007; Park et al. 2010). It is further characterized by weak vertical stratification of water properties, and is understood to be strongly influenced by Weddell Sea shelf water ventilating the deep water through the interactions of the ACC and the Weddell Gyre (Whitworth & Nowlin 1987).
Southern elephant seals form an integral part of the large Southern Ocean ecosystem and are considered major consumers, given their circumpolar distribution, large size and comparatively large population numbers. The diet of this species is poorly known, but likely consists mostly of squid and/or myctophid fish (Daneri et al. 2000; Daneri & Carlini 2002; Cherel et al. 2009; Newland et al. 2011). Individual specialization in foraging strategies and diets is thought to be pronounced (Hückstädt et al. 2012), although plasticity in foraging behaviour of individuals has also been reported for the species (e.g., Biuw et al. 2010; McIntyre, Ansorge et al. 2011). Southern elephant seals show changes in behaviour that are related, perhaps indirectly, to changes in physical oceanographic properties. For instance, Bestley et al. (2013) demonstrated that some juvenile male and adult female seals are more likely to switch to resident movement states in colder waters along the East Antarctic shelf area. Here they display relatively short bottom times during dives, although the resident movement states imply increased foraging. McIntyre, Ansorge et al. (2011) described a slightly different relationship for elephant seals from Marion Island, which dive to shallower depths and spend more time during the bottom phases of dives when foraging in colder waters.
Most studies of at-sea behaviour of southern elephant seals focussed on females and sub-adult males (e.g., Bailleul et al. 2008; McIntyre, Bornemann et al. 2011; Muelbert et al. 2013), and studies incorporating the behaviour of adult males are comparatively rare. Published accounts of adult male behaviour include dive behaviour of elephant seals from Macquarie Island (Hindell et al. 1991), Patagonia (Campagna et al. 1999), Marion Island (McIntyre et al. 2012) and King George Island (Isla 25 de Mayo) (Tosh et al. 2009; James et al. 2012). James et al. (2012) assessed differences in behaviour between small samples of adult males from two different populations. No other descriptive published accounts of the at-sea behaviour of adult male southern elephant seals from the Antarctic Peninsula region exist. Elephant seals (of diverse sex and age classes) from this region are known to target the western Antarctic Peninsula (McConnell et al. 1992; Bornemann et al. 2000; Hückstädt et al. 2012; Muelbert et al. 2013), areas in the immediate vicinity of King George Island around the northern tip of the Antarctic Peninsula (Tosh et al. 2009), as well as the Weddell Sea (Tosh et al. 2009). Here we report results from a study of the at-sea behaviour of 11 adult male southern elephant seals tracked from King George Island. We also illustrate, for the first time, the influences of a suite of environmental parameters on their dive behaviour and speed of travel.
Methods
We deployed 15 satellite-relay data loggers (SRDLs; Sea Mammal Research Unit, University of St. Andrews, Scotland) on post-moult adult male southern elephant seals hauled out at King George Island between March and April 2010. Ten devices were new conductivity–temperature–depth (CTD)–SRDLs capable of recording and transmitting temperature data with accuracy better than 0.005°C and salinity measures with accuracy better than 0.02 (Boehme et al. 2009), while five devices were refurbished CTD–SRDLs previously deployed on elephant seals elsewhere and re-fitted with new batteries. Visual comparison of temperature profiles prior to analyses confirmed that no systematic bias occurred in the temperature data of the re-batteried tags (see Supplementary Figs. S1, S2).
Seals were immobilized by remote intra-muscular injection of estimated dosages of tiletamine-zolazepam (Zoletil) between 0.5 and 0.69 mg/kg (mean: 0.63 mg/kg), and anaesthesia maintained by additional injections of ketamine as detailed in Bornemann et al. (2013). Post-hoc calculations of body weight (see below) revealed general overestimation of body mass and that the administered dosages were between 0.46 and 1.01 mg/kg (mean: 0.71 mg/kg). Four seals displayed periods of apnoea between 10 min and >25 min during the immobilization period, of which one seal required intervention to stimulate spontaneous respiration (see Bornemann et al. 2013 for further details). Standard lengths of seals in ventral recumbence were measured. Weight estimates for each tagged seal were calculated at deployment, using the photogrammetric method detailed in de Bruyn et al. (2009). We performed a linear regression of body weight (calculated) against standard length (measured) in order to obtain estimates of body condition for each seal at deployment (Schulte-Hostedde et al. 2005). All dive, track, temperature and associated meta-data are available via the PANGAEA Data Publisher for Earth & Environmental Science at http://doi.org/10.1594/PANGAEA.821555.
Tracks
Location estimates were obtained via the Service Argos satellite based system, using their least squares algorithm for calculating positions. Locations were most often assigned a location quality class of 0 (28% of the location estimates) by Service Argos, while locations of class A, B and Z made up a total of 47% of all location estimates. The accuracy of classes 0 and B locations is estimated between 5 and 10 km, while the remaining classes are likely to be accurate to approximately 2 km (Boyd & Brightsmith 2013). Track data were filtered based on assumed maximum swim speeds and turning angles (Freitas et al. 2008) as detailed in McIntyre, Bornemann et al. (2011). While not quantified, we expected the filtering process to result in significantly higher accuracy locations allowing for reasonably large-scale associations of environmental data with the available behavioural data (Kuhn et al. 2009). Filtered tracks were illustrated using ArcGIS 10.1 software. A travelling speed (m/s) was assigned to each retained location, based on time and great circle distance differences between that point and the preceding location—calculated using the adehabitatLT package (Calenge 2006) in the R environment—and assuming that seals swam at consistent speeds between location points retained after applying the filter described above.
Dives
Estimates of individual dive locations were provided by the manufacturers (Sea Mammal Research Unit, University of St. Andrews) and based on interpolated locations from position estimates provided by Service Argos after removing erroneous locations based on estimated maximum swim speeds of elephant seals (McConnell et al. 1992). Individual dives were labelled as having either occurred during the day, at night or during twilight (periods within 30 min of the local sunrise and sunset times), based on local time values of individual dives and local times of sunrise and sunset, calculated using the maptools package in the R environment (Lewin-Koh & Bivand 2012).
Data of individual dives consisted of four abstracted time/depth points representing the maximum dive depth and three points of greatest inflection. These data points were identified (from high resolution time/depth profiles recorded at 4 s intervals) onboard the devices using a broken stick algorithm prior to transmission (see Fedak et al. 2002 for further details). Elephant seals are known to increase time spent during the bottom phases of dives when evidently encountering prey items, as measured by acceleration loggers (Gallon et al. 2013). In order to obtain reasonable estimates of bottom time (time spent within the deepest 20% of each dive), we generated three additional time/depth points between each transmitted time/depth point, assuming constant swimming speeds and angles of ascent/descent between transmitted points, as described by McIntyre et al. (2010). This resulted in a total number of 21 time-depth points associated with each dive. We then used multivariate linear regressions to quantify the relationship between maximum dive depth, dive duration and bottom time for each track (separately for day and night dives) (Bailleul et al. 2008). Residuals from the regression were then used to identify dives of increased “forage effort”, based on above-average amounts of time spent at the bottoms of dives for particular dive depths and durations. Bottom time residuals as an indicator of foraging effort was previously used by a number of authors studying both elephant and Weddell (Leptonychotes weddellii) seals (Bailleul et al. 2008; McIntyre, Ansorge et al. 2011; McIntyre, Bornemann et al. 2011; Heerah et al. 2013; McIntyre et al. 2013). While Dragon et al. (2012) reported inconclusive relationships between bottom time residuals and evident increased foraging (negative bottom time residuals for deeper “active” dives and positive bottom time residuals for shallower “active” dives) in southern elephant seals, the results reported by Gallon et al. (2013) and Robinson et al. (2010) lend further support to this method.
Environmental variables
Seafloor depth estimates for each dive location were extracted from the ETOPO1 1 arc-minute global relief model from the National Oceanic and Atmospheric Administration’s National Geophysical Data Centre. Sea-ice concentrations for each location estimate were obtained from daily Advanced Microwave Scanning Radiometer (AMSR-E) imagery at a spatial resolution of 6.25 km (Spreen et al. 2008). In situ temperature records were recorded and transmitted by the CTD-SRDLs. Profiles were inspected visually using the Ocean Data View software package (Schlitzer 2002) for unrealistic temperature values (≤2.5°C or >7°C). No unrealistic values were evident in the data set and all transmitted temperature profiles were used for further analyses. For each of the temperature profiles, we calculated the maximum temperature value within the profile (Tmax), the depth of Tmax (Tmax.depth) and the difference between the maximum and minimum temperatures recorded (Tdiff).
Environmental influences on dive behaviour
We used a series of linear mixed effects models to identify the relative influences of environmental variables on dive depth (DDEP), dive duration (DDUR), travel speed (SP) and relative amount of time spent at the bottom of dives, as indicated by bottom time residuals (BTres). Transmitted dive and temperature profiles do not necessarily correspond in time or space due to the method of temporary storage of profiles onboard the tags prior to transmission (Boehme et al. 2009), and are limited to a maximum number of four profiles per day. In situ temperature characteristics were therefore attributed to transmitted dive profiles and speed locations, based on the closest CTD profile received in time. Dive profiles and location points with no associated CTD profiles successfully retrieved within a time period of 24 hrs around the location or dive profile were discarded for the mixed model analyses (but retained for descriptive statistics; see Table 1). Time differences between dive profiles and CTD profiles averaged 319±374 min and between speed location points and CTD profiles 254±338 min.
Our starting full models were:
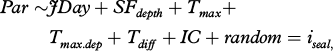
where Par = either DDEP, DDUR, SP or BTres; JDay=Julian day; SFdepth=seafloor depth (m); Tmax=maximum temperature (°C); Tmax.dep=depth of Tmax (m); Tdiff=temperature difference between maximum and minimum temperature (°C); IC=ice concentration (%); and iseal=individual seal (random term).
Table 1 Summary of dive statistics obtained for male southern elephant seals tagged at King George Island.
| Animal |
Length (cm) |
Weight estimate (kg) |
Condition residual |
Dive
(n) |
Dive depth (m) |
Max. depth (m) |
Dive duration (min) |
Max. duration (min) |
Surface duration (min) |
| J496 |
390 |
1241 |
−1.55 |
5859 |
632±362 |
2149 |
29.6±10.7 |
67.3 |
3.7±1 |
| J497 |
386 |
1913 |
2.17 |
5131 |
738±430 |
2389 |
34.5±11.2 |
88.3 |
3.5±1 |
| J498 |
381 |
1325 |
−0.56 |
5664 |
294±113 |
2149 |
24.4±9.8 |
77.3 |
2.8±1 |
| J500 |
430 |
1967 |
−0.38 |
3281 |
404±110 |
513 |
21.7±6.9 |
71.3 |
3.1±0.8 |
| J501 |
409 |
1950 |
0.86 |
4566 |
515±293 |
1589 |
35.9±14 |
95.3 |
3.2±1.1 |
| J502 |
380 |
1316 |
−0.54 |
7316 |
215±99 |
1129 |
24±9.6 |
78.3 |
2.7±1.1 |
| J503 |
378 |
1465 |
0.38 |
6719 |
555±368 |
2089 |
26.7±11.4 |
95.3 |
3.4±1.3 |
| J504 |
400 |
1462 |
−1.03 |
5558 |
321±105 |
2149 |
19.2±7.6 |
72.3 |
3.8±1.6 |
| J505 |
421 |
2064 |
0.69 |
4778 |
325±111 |
714 |
23.3±7.6 |
60.3 |
3±0.7 |
| Jr515 |
485 |
2694 |
−0.24 |
5493 |
188±78 |
2169 |
27.4±9.1 |
66.3 |
3.2±0.9 |
| Jr674 |
426 |
2010 |
0.1 |
5445 |
561±289 |
2349 |
27.7±9.7 |
82.3 |
2.9±0.8 |
Initial models consisted of all fixed effects. Since models mostly displayed significant temporal autocorrelation, we fitted autoregressive correlation functions (Pinheiro & Bates 2004). All possible combinations of fixed variables were then compared in order to select the most parsimonious models. Model selection was undertaken based on maximum likelihood and using second-order Akaike Information Criterion (AICc) and corresponding AIC weights to select the most parsimonious models (Burnham & Anderson 2002). After model selection, final models were run using restricted maximum likelihood (Bolker et al. 2008). We used a number of different plot types to assess model fits (Pinheiro & Bates 2004). Variance component analyses were carried out on all final models to estimate variation explained by the random term.
All analyses were undertaken in the R statistical environment (version 2.15.2; R Core Team 2012). We used the nlme package (Pinheiro et al. 2012) for mixed effect model analyses. Unless otherwise stated, mean values±SD are reported. Statistical significance was set at p≤0.05.
Results
Data were retained from 11 deployments after four CTD-SRDLs stopped transmitting prematurely. Six of the devices successfully transmitted information throughout the post-moult migrations of tagged seals, while the remaining five SRDLs included here stopped transmitting whilst the seals were at sea, but remained functional long enough to provide useful dive and track information (min. 74 days). Body mass estimates of tracked seals varied between 1241 and 2694 kg and standard body lengths between 378 and 485 cm (Table 1). For the 11 seals, 53 957 dive profiles and 5486 CTD profiles were recorded.
Foraging areas and dive behaviour
Tracked seals mostly foraged in areas of comparatively shallow bathymetry along the Scotia shelf during the post-moult trip, before heading to South Georgia Island for the breeding season haul-out (Fig. 1). Three seals (J504, J505, J498) targeted the South Orkney Plateau before either returning to South Georgia Island (J498) for the breeding haul-out or tags ceasing transmissions (J504, J505; Fig. 2). These three seals all experienced water conditions characterized by cold surface water (ca. −2°C) and slightly warmer sub-surface temperatures, and characteristic of water masses between the Antarctic Divergence and the Antarctic Slope Front (Park et al. 1998) (Fig. 3). They predominantly performed dives to the seafloor (e.g., J505; Fig. 5) at depths in the region of 300 m (Table 1). Travelling phases between King George Island, the South Orkney Plateau and South Georgia Island were characterized by comparatively short periods of time spent during bottom phases of dives.
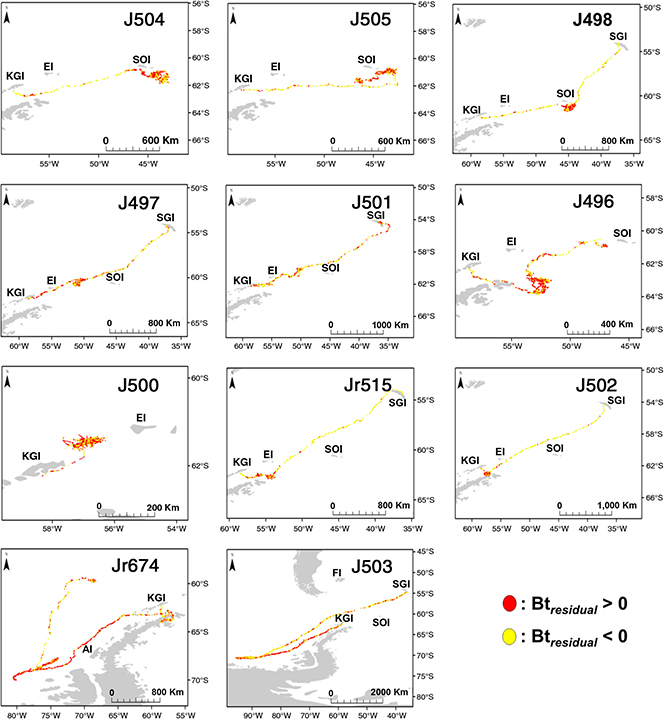
Fig. 1
Dive locations of individual seals tracked from King George Island, indicating daily averaged positions of dives with increased bottom times (BTresidual
>0) and dives with decreased bottom times (BTresidual
<0). Place names are abbreviated as follows: King George Island (KGI); Elephant Island (EI); South Georgia Island (SGI); Falkland Islands (Islas Malvinas; FI); South Orkney Islands (SOI); Adelaide Islands (AI).
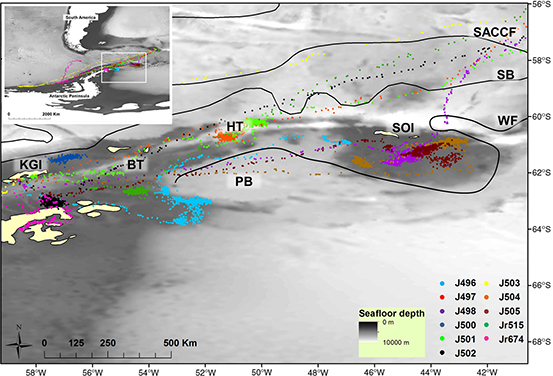
Fig. 2
Map of the Scotia Sea area utilized by seals in this study. Approximate locations of the boundary fronts of the Weddell–Scotia Confluence are indicated by black lines. Positions of the boundary fronts were estimated from Heywood et al. (2004) and Orsi et al. (1995) and as displayed by Meredith et al. (2011). The inset shows the distribution of all seals tracked in this study and highlights the area displayed in the main image. The following features and place names are abbreviated: Southern Antarctic Circumpolar Current Front (SACCF); southern boundary of the Antarctic Circumpolar Current (SB); Weddell Front (WF); King George Island (KGI); Bransfield Trough (BT); Hespérides Trough (HT); Powell Basin (PB); South Orkney Islands (SOI).
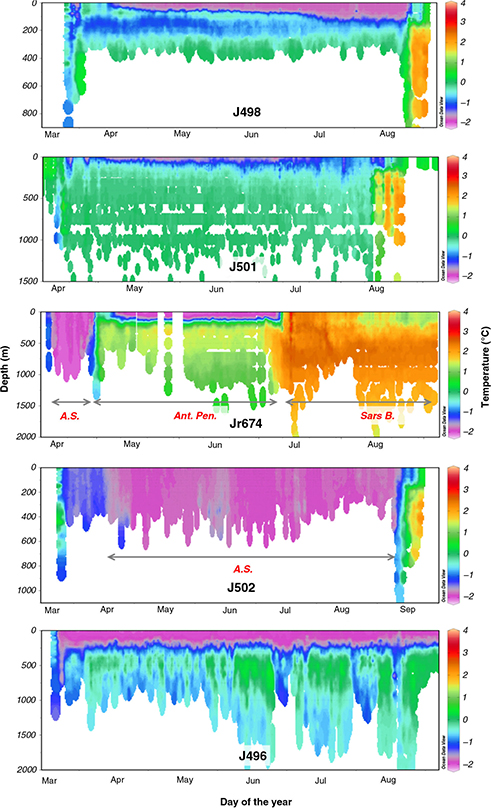
Fig. 3
Temperature collected by selected seals illustrating a variety of environments encountered. The following terms are abbreviated: Antarctic Shelf (A.S.); Antarctic Peninsula (Ant. Pen.); Sars Bank (Sars B.).
Two seals (J497, J501) targeted areas on different sides of the Hespérides Trough, showing increased foraging effort in these areas, before also returning to South Georgia Island for the breeding haul-out (Fig. 2). Water temperatures here were also characteristic of the area between the Antarctic Divergence and Antarctic Slope Front (e.g., J501; Fig. 3). These seals both tended to perform pelagic as well as benthic dives, and dived to depths mostly deeper than 500 m (Table 1).
Two seals, J503 and Jr674, travelled along the western side of the Antarctic Peninsula towards the Bellingshausen Sea. Jr674 first spent a brief period of time on the Antarctic Continental Shelf, as evidenced by water temperature profiles characteristic of this area south of the Antarctic Slope Front (Park et al. 1998; Bailleul, Carrassin, Monestiez et al. 2007; Fig. 3), before travelling further along the Antarctic Peninsula. Both of these seals increased their foraging effort on the southward (“outbound” from King George Island) part of their migrations, and displayed less foraging effort on the return phases. One of these animals (J503) returned along the peninsula, but slightly further offshore, to South Georgia Island. Jr674 did not travel as far along the Antarctic Peninsula before heading offshore and travelling to the Sars Bank, where the tag stopped transmitting. Both seals that travelled towards the Bellingshausen Sea performed a variety of dives that were mostly in close proximity to the seafloor at depths of approximately 550 m, but switched to pelagic dive strategies when moving away from the peninsula (e.g., Jr674; Fig. 4).
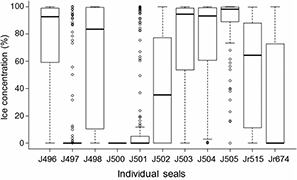
Fig. 4
Box and whisker plot of daily averaged sea-ice concentrations of the dive locations of tracked seals in this study. The bold lines indicate medians, boxes indicate 25th and 75th percentiles, individual points are outliers and whiskers indicate 1.5 times the interquartile range, or the maximum values (when there are no outliers).
The four remaining seals all targeted various areas in close proximity to King George Island. J500 moved to an area directly north of King George Island, adjacent to the South Shetland Trough and remained in this area until transmissions ceased from this device 74 days after departure. Both Jr515 and J502 crossed the Bransfield Strait and foraged in areas adjacent to Bransfield Island, before returning to South Georgia Island for the breeding haul-out. Water temperatures here were also characteristic of Antarctic Shelf waters, and showed no vertical stratification (e.g., J502; Fig. 3). These two seals also showed clear decreases in foraging effort on the travelling phases towards South Georgia Island. J496 also crossed the Bransfield Strait, but focussed its foraging in an area closer to the edge of the Powell Basin, before also returning to South Georgia Island along the edge of the Powell Basin. Areas targeted by this seal had water properties also consistent with the inter-frontal area between the Antarctic Divergence and Antarctic Slope Front (J496; Fig. 3). Three of the four seals that foraged close to King George Island (J500, J502, Jr515) also mostly performed dives to the seafloor—J502 and Jr515 to depths in the region of 200 m and J500 to deeper seafloor depths of approximately 400 m (Table 1).
Seals displayed much variation in how much time they spent in areas of high sea-ice concentrations. Six of the seals spent most of their foraging migrations in areas of very high sea-ice concentrations—up to 100% (e.g., J505; Fig. 5), while some individuals foraged almost entirely in ice-free environments (e.g., J497; Fig. 5). There were no evident trends in relationships between length, mass or condition of the seals and sea-ice concentrations they foraged in (Supplementary Fig. S3). Also, body size and condition variables (length, mass and condition) of tracked seals did not show any obvious relationships with dive depths, dive durations or time spent at the bottoms of dives (time or percentage) (Supplementary Fig. S4).
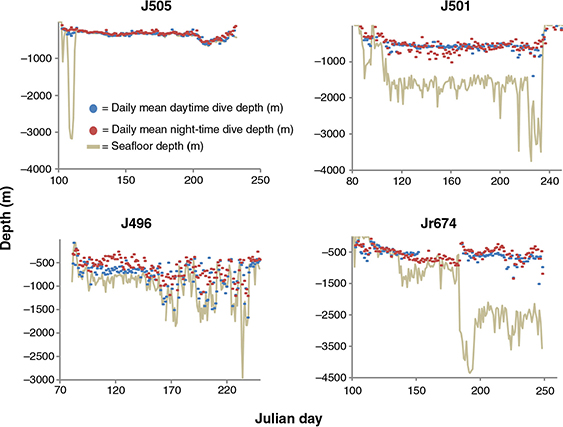
Fig. 5
Time-series plots of daily averaged day- and night-time dive depths of four tracked seals in relation to the seafloor depth illustrating different foraging strategies, including virtually exclusive benthic diving (J505), mostly pelagic diving (J501) and combinations of different strategies (J496, Jr674).
Dive depths
The full model containing all environmental parameters provided the best fit to daytime dive depths (Table 2). Variables showing significant associations with dive depths included SFdepth, Tmax.dep, Tdiff and IC (Table 3). The associations of these variables were mostly positive, except for IC, indicating slightly shallower dive depths in higher sea-ice concentrations. The largest coefficient associated with a fixed effect was the value of 1.2 associated with Tdiff, suggesting that seals increased their dive depths in areas with greater temperature stratification in the water column. The best model for night-time dive depths excluded SFdepth, but included all other fixed effects (Table 2). Significant associations with night-time dive depths were detected from JDay, Tmax.dep,Tdiff and IC. The final model used to explain dive depths occurring during twilight periods included only SFdepth, Tmax.dep and Tdiff as fixed effects, with all of these showing statistically significant associations with dive depth (Table 2).
Table 2 Summary of best fit linear mixed models used in this study.
| Behaviour |
Model |
Δ AICca
|
Random effect |
| DDEPDAY
|
sqrt(DDEPDAY)~ JDay+SFdepth+Tmax+Tmax.dep+Tdiff+IC
|
0 |
27.4 |
| DDEPNIGHT
|
sqrt(DDEPNIGHT)~ JDay+Tmax+Tmax.dep+Tdiff+IC
|
1.94 |
23.1 |
| DDEPTWILIGHT
|
sqrt(DDEPTWILIGHT)~ SFdepth+Tmax.dep+Tdiff
|
5.17 |
15.2 |
| DDURDAY
|
DDURDAY ~ JDay+SFdepth+Tmax+Tmax.dep+Tdiff+IC
|
0 |
28.9 |
| DDURNIGHT
|
DDURNIGHT ~ JDay+SFdepth+Tmax.dep+Tdiff+IC
|
2 |
19.9 |
| DDURTWILIGHT
|
DDURTWILIGHT ~ JDay+SFdepth+Tmax+Tmax.dep+Tdiff
|
1.4 |
17.5 |
| SPDAY
|
sqrt(SPDAY) ~ JDay+SFdepth+Tmax+Tmax.dep+IC
|
1.81 |
4.2 |
| SPNIGHT
|
sqrt(SPNIGHT) ~ JDay+SFdepth+Tmax+Tmax.dep+Tdiff+IC
|
0 |
3.6 |
| SPTWILIGHT
|
sqrt(SPTWILIGHT) ~ JDay+SFdepth+Tmax.dep+Tdiff+IC
|
1.96 |
2.9 |
| BTDAY
|
BTDAY ~ JDay+SFdepth+Tmax+Tmax.dep+Tdiff+IC
|
0 |
0.8 |
| BTNIGHT
|
BTNIGHT ~ JDay+SFdepth+Tdiff+IC
|
2.17 |
0.7 |
| BTTWILIGHT
|
BTTWILIGHT ~ JDay+SFdepth+Tmax
|
16.41 |
2.9 |
| aDifference in second-order Akaike Information Criterion value between final model displayed here and full starting model. |
Table 3 F-test results indicating significant fixed effects on all final models. Boldface values are significant at the 0.05 level.
| |
Day |
Night |
Twilight |
| Parameter |
Fixed effect |
Coeff. |
F
|
df |
p
|
Fixed effect |
Coeff. |
F
|
df |
p
|
Fixed effect |
Coeff. |
F
|
df |
p
|
| DDEP |
(Intercept) |
15.58 |
16.66 |
1,5 696 |
<0.001 |
(Intercept) |
14.849 |
14.41 |
1,15 663 |
<0.001 |
(Intercept) |
13.669 |
19.2 |
1,679 |
<0.001 |
| |
JDay
|
0.003 |
1.16 |
1,5 696 |
0.248 |
JDay
|
0.006
|
2.29
|
1,15 663
|
0.022
|
SFdepth
|
0.001
|
3.17
|
1,679
|
0.002
|
| |
SFdepth
|
0.001
|
6.48
|
1,5 696
|
<0.001
|
Tmax
|
0.189 |
0.91 |
1,15 663 |
0.363 |
Tmax.dep
|
0.01
|
9.71
|
1,679
|
<0.001
|
| |
Tmax
|
0.38 |
1.58 |
1,5 696 |
0.115 |
Tmax.dep
|
0.004
|
11.12
|
1,15 663
|
<0.001
|
Tdiff
|
1.47
|
5.25
|
1,679
|
<0.001
|
| |
Tmax.dep
|
0.006
|
11.54
|
1,5 696
|
<0.001
|
Tdiff
|
2.189
|
11.86
|
1,15 663
|
<0.001
|
|
|
|
|
|
| |
Tdiff
|
1.166
|
5.18
|
1,5 696
|
<0.001
|
IC
|
−0.01
|
−3.93
|
1,15 663
|
0.001
|
|
|
|
|
|
| |
IC
|
−0.007
|
−2.13
|
1,5 696
|
0.033
|
|
|
|
|
|
|
|
|
|
|
| DDUR |
(Intercept) |
816.055 |
8.13 |
1,5 969 |
<0.001 |
(Intercept) |
881.13 |
10.17 |
1,15 663 |
<0.001 |
(Intercept) |
815.72 |
6.66 |
1,677 |
<0.001 |
| |
JDay
|
3.035
|
10.95
|
1,5 696
|
<0.001
|
JDay
|
3.24
|
14.34
|
1,15 663
|
<0.001
|
JDay
|
2.535
|
4.81
|
1,677
|
<0.001
|
| |
SFdepth
|
0.122
|
9.13
|
1,5 696
|
<0.001
|
SFdepth
|
0.04
|
3.67
|
1,15 663
|
<0.001
|
SFdepth
|
0.149
|
4.43
|
1,677
|
<0.001
|
| |
Tmax
|
−43.082 |
−1.71 |
1,5 969 |
0.088 |
Tmax.dep
|
0.348
|
9.56
|
1,15 663
|
<0.001
|
Tmax
|
−44.382 |
−0.95 |
1,677 |
0.342 |
| |
Tmax.dep
|
0.372
|
6.98
|
1,5 969
|
<0.001
|
Tdiff
|
84.425
|
6.26
|
1,15 663
|
<0.001
|
Tmax.dep
|
0.665
|
5.25
|
1,677
|
<0.001
|
| |
Tdiff
|
138.57
|
5.87
|
1,5 969
|
<0.001
|
IC
|
−1.28
|
−5.33
|
1,15 663
|
<0.001
|
Tdiff
|
86.789
|
1.98
|
1,677
|
0.048
|
| |
IC
|
−1.322
|
−3.88
|
1,5 969
|
<0.001
|
|
|
|
|
|
|
|
|
|
|
| SP |
(Intercept) |
0.906 |
23.47 |
1,5 593 |
<0.001 |
(Intercept) |
0.923 |
28.09 |
1,15 427 |
<0.001 |
(Intercept) |
0.905 |
13.71 |
1,671 |
<0.001 |
| |
JDay
|
<−0.001
|
−2.51
|
1,5 593
|
0.012
|
JDay
|
<−0.001
|
−7.37
|
1,15 427
|
<0.001
|
JDay
|
−0.001
|
−3.77
|
1,671
|
<0.001
|
| |
SFdepth
|
<−0.001
|
5.89
|
1,5 593
|
<0.001
|
SFdepth
|
<0.001
|
11.44
|
1,15 427
|
<0.001
|
SFdepth
|
<0.001
|
4.18
|
1,671
|
<0.001
|
| |
Tmax
|
0.06
|
4.49
|
1,5 593
|
<0.001
|
Tmax
|
−0.015 |
−1.32 |
1,15 427 |
0.186 |
Tmax.dep
|
<−0.001 |
−1.84 |
1,671 |
0.067 |
| |
Tmax.dep
|
<−0.001 |
−1.25 |
1,5 593 |
0.213 |
Tmax.dep
|
<−0.001
|
−2.42
|
1,15 427
|
0.016
|
Tdiff
|
0.071
|
3.08
|
1,671
|
0.002
|
| |
IC
|
−0.002
|
−7.48
|
1,5 593
|
<0.001
|
Tdiff
|
0.024
|
2.55
|
1,15 427
|
0.012
|
IC
|
<−0.001
|
−2.51
|
1,671
|
0.012
|
| |
|
|
|
|
|
IC
|
−0.002
|
−10.62
|
1,15 427
|
<0.001
|
|
|
|
|
|
| BT |
(Intercept) |
0.284 |
3.67 |
1,5 696 |
<0.001 |
(Intercept) |
0.277 |
5.72 |
1,15 664 |
<0.001 |
(Intercept) |
0.397 |
2.89 |
1,679 |
0.004 |
| |
JDay
|
−0.001
|
−3.96
|
1,5 696
|
<0.001
|
JDay
|
−0.002
|
−5.35
|
1,15 664
|
<0.001
|
JDay
|
−0.001 |
−1.49 |
1,679 |
0.136 |
| |
SFdepth
|
<−0.001
|
−4.36
|
1,5 696
|
<0.001
|
SFdepth
|
<−0.001
|
−7.68
|
1,15 664
|
<0.001
|
SFdepth
|
<−0.001
|
−2.5
|
1,679
|
0.012
|
| |
Tmax
|
−0.086
|
−2.57
|
1,5 696
|
0.01
|
Tdiff
|
0.044
|
3.37
|
1,15 664
|
<0.001
|
Tmax
|
0.147
|
2.78
|
1,679
|
0.006
|
| |
Tmax.dep
|
<−0.001 |
−1.54 |
1,5 696 |
0.12 |
IC
|
<−0.001 |
−1.89 |
1,15 664 |
0.06 |
|
|
|
|
|
| |
Tdiff
|
0.18
|
5.24
|
1,5 696
|
<0.001
|
|
|
|
|
|
|
|
|
|
|
| |
IC
|
<−0.001 |
−1.38 |
1,5 696 |
0.168 |
|
|
|
|
|
|
|
|
|
|
Individual variability explained 27.4% of the variance in dive depths occurring in daytime, 23.1% in night-time and 15.2% in twilight. Overall temperature difference within profiles (Tdiff) showed the strongest likely influence on both day- and night-time dive depths, dives consistently increasing in depth as Tdiff increased. The influence of Tmax.dep was similar between day- and night-time dives, and indicated a slight increase in dive depths, when the Tmax.dep was located deeper. Increases in SFdepth. were associated with slightly increased dive depths for both daytime and twilight dives. Ice concentration (IC) showed statistically significant effects on daytime and night-time dive depths, and seals evidently slightly decreased their dive depths in areas with higher sea-ice concentrations.
Dive durations
Daytime dive durations were best explained by a full model that included all fixed effects, while the most parsimonious model for night-time dive durations was similar, but excluded Tmax
, and the best model for twilight dive durations included all fixed effects, except IC (Table 2). Individual variation explained 28.9% of the daytime, 19.9% of the night-time and 17.5% of twilight variance in dive durations.
Comparatively large influences were detected from Tdiff for dives performed during all times of day (Table 3): dives becoming longer as Tdiff increased. Dives further tended to get longer as migrations progressed, as evidenced by the positive relationship of dive durations with JDay (Table 3). We found evidence for a significant correlation between JDay and Tdiff for dives performed during the day, but not during other times. The depth of Tmax (Tmax.dep) was further positively related to dive durations, and longer dives were recorded in water where the Tmax.dep was deeper. Dive durations were further positively related to SFdepth, seals tending to undertake longer dives when in deeper water, although dives in deeper water were mostly not to the seafloor (Fig. 3). Sea-ice concentrations (IC) negatively affected durations of dives performed during the day and at night, and seals performed slightly shorter dives when in areas with higher IC.
Bottom time residuals
The relative amounts of time spent during the bottom phases of dives, as indicated by the bottom time residuals (BTres), during daytime dives were best explained by the full model containing all fixed effects, while night-time bottom times were best explained by a model containing JDay, SFdepth, Tdiff and IC as fixed effects (Table 2). Twilight bottom time residuals were best explained by a model containing only JDay, SFdepth and Tmax as fixed effects. Individual variation explained more of the twilight model variance (2.9%) than the daytime (0.9%) and night-time (0.7%) model variance. Seafloor depth consistently influenced the amount of time spent during bottom phases, and seals spent comparatively more time during the bottom phases of dives in areas with shallower seafloors (Table 3). Coefficient sizes suggest that Tdiff had a greater influence on day- and night-time bottom times, with seals increasing bottom times in areas with increased temperature stratification of the water column.
Travelling speed
The best models to explain travelling speed included all fixed effects for night-time travel, and all effects, except Tdiff for daytime travel (Table 2). Twilight speed was best explained by a model that contained all fixed effects, except for Tmax. Individual variation explained small proportions of model variance: 0.8% for daytime, 0.7% for night-time and 2.9% for twilight speed.
Bathymetry (SFdepth) and sea-ice concentration (IC) showed small, but significant and consistent influences on travelling speed, indicating that seals tended to travel slightly slower through areas of shallower water, as well as areas with increased sea-ice cover (Table 3). The depth of Tmax (Tmax.dep) had consistently small negative influences on dive bottom times, although this relationship was only statistically significant for night-time dives.
Discussion
These male elephant seals tracked from King George Island mostly focussed their foraging efforts in the vicinity of the Weddell–Scotia Confluence between King George Island and the South Orkney Islands, before migrating to South Georgia Island for the breeding season haul-out. These movements differed to those reported by Tosh et al. (2009) for male seals instrumented at the same locality. Some of those males travelled into the Weddell Sea where they clearly focussed their foraging efforts around the Filchner Trough outflow. Such differences between the studies may be indicative of any of several factors, including differing natality or even interannual differences in movements of animals from this population. Tracked seals were not previously known animals (no permanent markings from tagging efforts at any of the breeding colonies around the sub-Antarctic). Since southern elephant seals are known to display high levels of site fidelity to their natal sites (Lewis et al. 1996; Hofmeyr et al. 2012), and these animals travelled to South Georgia Island for the breeding season haul-out, it is probable that they were actually born on South Georgia Island and use King George Island as a moult haul-out site. Such a strategy was also observed for seals tracked from Elephant Island, which returned to South Georgia Island for the breeding season haul-out (Muelbert et al. 2013). In contrast, two out of the three seals that foraged deep in the Weddell Sea in the Tosh et al. (2009) study were known individuals, either from scarring patterns or from being branded on King George Island as weaned pups (Horst Bornemann, personnel observation). The differences observed in movements between the two studies may also be due to variation in sea-ice conditions between the study years (potentially preventing movement of seals into the Weddell Sea in the recent study), or it may simply relate to individual variation in preferred migration routes.
Seals tended to target areas of comparatively shallow bathymetry (this study), but displayed some variation in dive strategies: most animals foraged benthically in relatively shallow water, but others pelagically in deep water, and some displayed combinations of benthic and pelagic dives. Nine of 11 tracked animals focussed their foraging efforts within the Scotia Sea ecosystem, while two seals travelled along the western Antarctic Peninsula shelf (Fig. 2).
Decreased travel speed and increased relative amounts of time spent during the bottom phases of dives were interpreted as indicating increased foraging effort in our study. While increased bottom times could equate to something other than foraging and may not be the best predictor of foraging effort (Dragon et al. 2012), a number of papers suggest that it is likely to correspond with increases in foraging effort (e.g., Robinson et al. 2010; Gallon et al. 2013). Travelling speed has further been shown to be a good indicator of foraging effort in elephant seals, with seals slowing travel speeds in areas of increased foraging (Robinson et al. 2010). Other assumptions we made in our study included no influence associated with potential mismatches in datasets associated with differences in spatial resolution of data sets or potential mismatches due to temporal differences between data sets (e.g., dive data vs. in situ temperature values). While it is difficult to account for the spatial error associated with the accuracy of Argos locations in our study (no higher accuracy devices, such as global positioning system tags, were deployed concurrently with the SRDLs), previous studies have illustrated that appropriately filtered Argos location data can be closely related to the spatial scale of remotely sensed data (e.g., Kuhn et al. 2009). The method we used to match dive data with in situ temperature data resulted in mismatches that were often in the region of 5 hrs. This mismatch could potentially have resulted in seal dive locations having occurred at substantial distances from the recorded CTD profiles, thereby introducing errors into the associations between dives and the temperature structure of the water column. However, seals in our study moved at an average speed of approximately 2.8 km/hr (0.76 m/s), suggesting that most dive locations would have been within 14 km of the closest CTD profile (although this distance was likely to be smaller, since the animals were expected to mostly not follow straight swimming trajectories). While difficult to quantify, mismatches at this scale were considered unlikely to introduce substantial bias into our results, since the areas mostly targeted by seals in our study are characterized by reasonably large areas of comparatively homogenous water masses (Whitworth et al. 1994).
Bathymetry associations with dive behaviour
Model outputs suggest that seals tended to dive for longer periods of time, but spend less time during the bottom phases of dives when travelling over areas of increased depth (SFdepth
)—conversely, increasing their bottom time, but diving for shorter absolute times over shallower areas. The shorter absolute dive times and longer bottom times in shallower areas suggest that dive durations were unlikely to be influenced by estimated aerobic dive limits, since similar or longer absolute dive times would be expected in areas where seals are presumably increasing forage effort and therefore likely maximizing time spent diving. Indeed, if assuming calculated aerobic dive limits (cADL) of between 40 and 50 min—based on the cADLs of adult male southern elephant seals of a similar size in Hindell et al. (1992)—it is clear that males in our study mostly dived well within their cADLs (Table 1), although all animals performed at least some dives exceeding such durations as well. Dives that exceeded 40 min (and were considered likely to approach or exceed cADLs of the tracked seals) totalled 11.8% of all dives recorded. These dives (≥40 min) generally occurred over deeper water than other dives (Wilcoxon test: W=10998717, p<0.001), lending further support to the suggestion that dives associated with increased forage effort were unlikely to be limited by aerobic dive limits in our study animals.
Furthermore, tracked seals tended to travel faster over deeper areas, than over areas of shallow bathymetry. Travelling speed is considered to be a good predictor of foraging effort in elephant seals, with faster travel speeds associated with decreased foraging and vice versa (Robinson et al. 2010). Increased bottom time is also positively related to likely prey encounters in southern elephant seals (Gallon et al. 2013). Our models showed generally good agreement of bathymetric influences on both these behavioural parameters.
Male southern elephant seals are known to often perform benthic dives in various parts of the Southern Ocean where the bathymetry is comparatively shallow (Hindell et al. 1991; Campagna et al. 1999; Campagna et al. 2007; McIntyre et al. 2012). Seafloor depth did not seem to influence many of the dive characteristics of male seals tracked from Marion Island (McIntyre, Ansorge et al. 2011) although James et al. (2012) showed that dive depths were partly explained by seafloor depth. The influence of seafloor depth on dive characteristics observed here is consistent with results reported for seals from nearby Elephant Island that also target areas of shallower bathymetry with gentler slopes (Muelbert et al. 2013).
Sea-ice associations with dive behaviour
Sea-ice concentrations were associated with day- and night-time behavioural parameters measured in our study, although these influences were not always statistically significant (Table 3). Seals tended to perform shallower and shorter dives in areas with higher ice concentrations, but increased the dive bottom times and decreased travelling speed. This may be a result of the seals foraging on the shallower continental shelf, within an area of increased sea-ice concentration. However, our models did not indicate any correlations between seafloor depth and sea-ice concentrations. Seals tracked in our study often spent substantial portions of their foraging migrations in areas with very high ice concentrations, similar to some of the male seals tracked by Tosh et al. (2009), as well as some adult females tracked by Bornemann et al. (2000). Muelbert et al. (2013) reported a relationship between the size of tracked male and female elephant seals and the time spent in areas of high sea-ice concentrations. They suggested that larger seals may be better able to cope with higher sea-ice concentrations and that younger seals are perhaps unable to exploit such environments. Similar findings were reported by Bailleul, Charrassin, Ezraty et al. (2007), suggesting that female seals avoided high sea-ice concentrations, while larger sub-adult males were able to exploit higher sea-ice concentrations. We found no clear relationship between the size (or condition) of adult male seals and the sea-ice concentrations of the areas that they foraged in (Fig. 3). However, our results suggest that areas of high sea-ice concentrations are likely to be attractive foraging grounds for southern elephant seals, given the increased foraging effort of our study animals. The lack of a relationship between size and sea-ice concentrations in our study is likely due to our sample only including comparatively large seals, all possibly capable of exploiting high ice concentration areas.
Water temperature influences on dive behaviour
Many of the seals in our sample concentrated their foraging efforts within the Weddell–Scotia Confluence (Fig. 2). This area is characterized by weakly stratified water that is comparatively homogeneous, colder, saltier and with higher O2 concentrations than water north and south of this region (Whitworth et al. 1994). When these seals left this area, and migrated to South Georgia Island, they encountered more stratified water masses in closer proximity to their haul-out location (Atkinson et al. 2001). Time-series plots of the water characteristics encountered by seals in our study clearly illustrate such differences in water properties encountered by seals when they moved from the Weddell–Scotia Confluence area towards South Georgia Island (e.g., J498, J501, J502; Fig. 3). While the seals encountered areas with very different water temperature properties, little seasonal changes in water temperatures at depth were apparent in the time-series profiles of seals that focussed their dive efforts within small areas for the majority of their migrations (e.g., J498, J501, J502; Fig. 3). These results agree well with those reported from a similar dataset by Meredith et al. (2011), who also illustrated a stable temperature regime at depth. Substantial individual variation was present in the water masses targeted by tracked seals, with two individuals focussing their foraging efforts in homogeneous cold waters associated with the continental shelf and others targeting areas with some stratification in temperature profiles. Interestingly, comparatively few seals in our study targeted waters associated with the continental shelf (n = 2). This is in contrast to results from Bailleul, Charrassin, Monestiez et al. (2007), who showed that male and female southern elephant seals tracked from the Kerguelen Islands mostly targeted the continental shelf waters.
Tracked seals in our study adjusted their dive depths and dive durations when swimming through areas with different temperature profiles, and also exhibited changes in the amount of time spent during the bottom phases of dives (Table 3). Seals that moved between different water masses consistently dived deeper and for longer, when foraging within water masses that showed increased differences between minimum and maximum temperatures of the dive profiles (Tdiff) (Table 3). Additionally, day- and night-time dives tended to be more “square-shaped” in terms of their time-depth profiles due to increased bottom times when seals were foraging in areas with increased Tdiff. This may indicate differences in the vertical depth distribution of prey species associated with differences in the temperature gradient of the water column—possibly suggesting aggregations of suitable prey at deeper depths in more stratified water masses, and more scattered distributions of prey in weakly stratified water masses (Takahashi et al. 2008). Similar relationships between dive parameters of Weddell seals and the temperature stratification of the water column were described recently by McIntyre et al. (2013).
The maximum temperature of water profiles (Tmax) only significantly influenced daytime and twilight bottom time residuals, as well as speed of travel during the daytime (Table 3), although it was included as a variable in the best fit models for dive depth (Table 2). The directions of influence are similar to those reported by McIntyre, Ansorge et al. (2011) for seals tagged at Marion Island, where seals dive deeper and display shorter bottom times in areas of higher water temperature. The influence of Tmax on behavioural parameters (in relation to other environmental parameters) was apparently smaller in this study compared to what was reported for the Marion Island elephant seals. Such differences in behavioural adjustments are possibly due to a combination of search strategy differences between seals from the two populations, as well as the actual water masses targeted for foraging. Male seals from Marion Island tend to display fewer areas of concentrated movement (although there are exceptions), spending less time in these and pursue pelagic strategies whereby they travel further distances away from the island (McIntyre et al. 2012). Seals from King George Island travel shorter distances and spend more time in a few areas of concentrated movement (Tosh et al. 2009; this study). Furthermore, much of the area utilized by seals from Marion Island is characterized by strong sub-surface temperature maxima and comparatively stronger vertical stratification in the water columns (Pollard et al. 2002), as compared with the more homogeneous water structure of the Weddell–Scotia Confluence utilized by the seals from King George Island. McIntyre, Ansorge et al. (2011) suggested that the elephant seals at Marion Island may experience negative impacts, having to dive deeper and/or shift their migrations polewards to follow prey distribution shifts as the Southern Ocean continues to warm. Our results here suggest that male elephant seals utilising the Weddell-Scotia Confluence are unlikely to face similar challenges, since they are (currently) evidently diving well within their physiological limits, and exhibiting much plasticity in foraging behaviours associated with different oceanographic conditions.
Southern elephant seals tagged at Bouvetøya (Bouvet Island) displayed dive depths that were often closely associated with the sub-surface temperature maximum depth of the water column, with seals sometimes evidently targeting prey occurring within this water depth layer (Biuw et al. 2010). While Tmax.depth did show consistent and statistically significant relationships with dive depths and dive durations of seals in our sample (Table 3), time-series plots did not illustrate any clear targeting of this water depth layer (e.g., J496; Fig. 6), although some seals did display some apparent concentration of dive effort in the general depth layer associated with the Tmax (e.g., J501; Fig. 6).
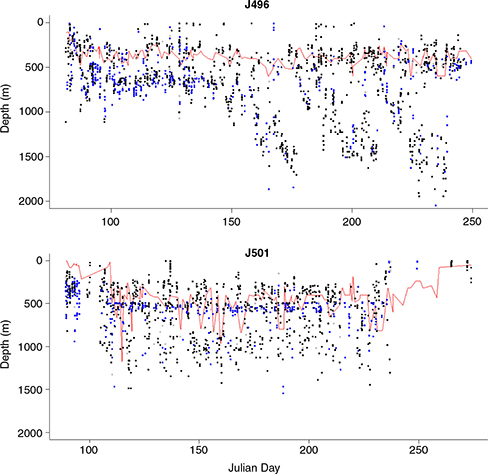
Fig. 6
Example time-series plots of dive depths in relation to Tmax.depth (red line) for two seals, J496 and J501. Blue dots indicate daytime dives and black dots night-time dives.
Conclusion
Male southern elephant seals tracked on their post-moult migrations from King George Island focussed their foraging efforts mostly within the Weddell–Scotia Confluence, while two animals foraged along the western Antarctic Peninsula shelf. The foraging behaviour of tracked seals was mostly influenced by bathymetry as well as sea ice, and seals increased their foraging effort in areas of shallower water and areas characterized by increased sea-ice concentrations. Water temperature structure also showed strong relationships with the differences in dive depth and dive durations of seals, as well as relative amounts of time spent during the bottom phases of dives—likely as a response to vertical changes in prey distribution associated with temperature. Compared to other studies, these results illustrate differences in behaviour of southern elephant seals utilizing different areas within the Southern Ocean (with differing associated hydrographic regimes). It further suggests that male elephant seals foraging in the Weddell–Scotia Arc and Antarctic Peninsula region are likely to be capable of adjusting behaviourally to potential changes in prey distribution associated with expected environmental changes—likely with lesser associated physiological costs, compared to elephant seals from Marion Island (McIntyre, Ansorge et al. 2011). However, future studies including year-round behavioural data, as well as higher resolution dive information associated with longer-term measures of fitness (e.g., survival and reproductive success), could add much to our understanding of potential consequences associated with behavioural patterns. Furthermore, while southern elephant seals often display behavioural plasticity in their at-sea behaviour, region-specific approaches to studying environmental influences on behavioural tendencies may allow for the identification of specific behavioural tendencies in different environments. Such information would further better inform the identification and management of relevant environments.
Acknowledgements
This paper is dedicated to the memory of late Alejandro Carlini who greatly facilitated elephant seal research at King George Island through a co-operation agreement between the Alfred Wegener Institute Helmholtz Center for Polar and Marine Research and the Argentine Antarctic Institute. The Alfred Wegener Institute for Polar and Marine Research provided financial and logistical support for the field campaign to deploy satellite tags. The immobilization of male southern elephant seals and the deployment of satellite transmitters within the Antarctic Specially Protected Area No. 132 at King George Island (Isla 25 de Mayo) were approved by the Argentine National Antarctic Program, Argentine Antarctic Institute, Buenos Aires, Argentina, and carried out pursuant to the Scientific Committee on Antarctic Research Code of Conduct for Animal Experiments. TM was supported by an International Climate Protection Fellowship from the Alexander von Humboldt Foundation during the data analyses and writing of this paper. We further wish to thank Mark Jessopp and three anonymous reviewers for their constructive comments on previous versions of this manuscript.
References
Atkinson
A.,
Siegel
V.,
Pakhomov
E.A.,
Jessopp
M.J.
&
Loeb
V. 2009.
A re-appraisal of the total biomass and annual production of Antarctic krill. Deep-Sea Research Part I 56,
727–740.
Publisher Full Text
Atkinson
A.,
Whitehouse
M.J.,
Priddle
J,
Cripps
G.C.,
Ward
P.
&
Brandon
M.A. 2001.
South Georgia, Antarctica: a productive, cold water pelagic ecosystem. Marine Ecology Progress Series 216,
279–308.
Publisher Full Text
Bailleul
F.,
Charrassin
J.-B.,
Ezraty
R.,
Girard-Ardhuin
F.,
McMahon
C.R.,
Field
I.C.
&
Guinet
C. 2007.
Southern elephant seals from Kerguelen Islands confronted by Antarctic sea ice. Changes in movements and diving behaviour. Deep-Sea Research Part II 54,
343–355.
Publisher Full Text
Bailleul
F.,
Charrassin
J.-B.,
Monestiez
P.,
Roquet
F.,
Biuw
M.
&
Guinet
C. 2007.
Successful foraging zones of southern elephant seals from the Kerguelen Islands in relation to oceanographic conditions. Philosophical Transactions of the Royal Society B 362,
2169–2181.
Publisher Full Text
Bailleul
F.,
Pinaud
D.,
Hindell
M.,
Charrassin
J.-B.
&
Guinet
C. 2008.
Assessment of scale-dependent foraging behaviour in southern elephant seals incorporating the vertical dimension: a development of the first passage time method. Journal of Animal Ecology 77,
948–957.
PubMed Abstract | Publisher Full Text
Bestley
S.,
Jonsen
I.D.,
Hindell
M.A.,
Guinet
C.
&
Charrassin
J.-B.
2013. Integrative modelling of animal movement: incorporating in situ habitat and behavioural information for a migratory marine predator. Proceedings of the Royal Society B 280, article no. 20122262, doi: 10.1098/rspb.2012.2262.
Biuw
M.,
Nøst
O.A.,
Stien
A.,
Zhou
Q.,
Lydersen
C.
&
Kovacs
K.M.
2010. Effects of hydrographic variability on the spatial, seasonal and diel diving patterns of southern elephant seals in the eastern Weddell Sea. PLoS One
5, e13816, doi: 10.1371/journal.pone.0013816.
Boehme
L.,
Lovell
P.,
Biuw
M.,
Roquet
F.,
Nicholson
J.,
Thorpe
S.E.,
Meredith
M.P.
&
Fedak
M. 2009.
Technical note: animal-borne CTD-satellite relay data loggers for real-time oceanographic data collection. Ocean Science 5,
685–695.
Publisher Full Text
Bolker
B.M.,
Brooks
M.E.,
Clark
C.J.,
Geange
S.W.,
Poulsen
J.R.,
Stevens
H.H.
&
White
J.-S.S. 2008.
Generalized linear mixed models: a practical guide for ecology and evolution. Trends in Ecology and Evolution 24,
127–135.
Publisher Full Text
Bornemann
H.,
de Bruyn
P.J.N.,
Reisinger
R.R.,
Kästner
S.,
Márquez
M.E.I.,
McIntyre
T.,
Bester
M.N.
&
Plötz
J. 2013.
Tiletamine/zolazepam immobilisation of adult post moult southern elephant seal males. Polar Biology 36,
1687–1692.
Publisher Full Text
Bornemann
H.,
Kreyscher
M.,
Ramdohr
S.,
Martin
T.,
Carlini
A.R.,
Sellmann
L.
&
Plötz
J. 2000.
Southern elephant seal movements and Antarctic sea ice. Antarctic Science 12,
3–15.
Publisher Full Text
Boyd
D.
&
Brightsmith
D.
2013. Error properties of Argos satellite telemetry locations using least squares and Kalman filtering. PLoS ONE
8, e63051, doi: 10.1371/journal.pone.0063051.PubMed Central Full Text
Burnham
K.P.
&
Anderson
D.R. 2002. Model selection and multimodel inference, a practical information–theoretic approach, 2nd edn. New York: Springer.
Calenge
C. 2006.
The package adehabitat for the R software, a tool for the analysis of space and habitat use by animals. Ecological Modelling 197,
516–519.
Publisher Full Text
Campagna
C.,
Fedak
M.
&
McConnell
B.J. 1999.
Post-breeding distribution and diving behavior of adult male southern elephant seals from Patagonia. Journal of Mammalogy 80,
1341–1352.
Publisher Full Text
Campagna
C.,
Piola
A.R.,
Rosa Marin
M.,
Lewis
M.,
Zajaczkovski
U.
&
Fernandez
T. 2007.
Deep divers in shallow seas: southern elephant seals on the Patagonian shelf. Deep-Sea Research Part I 54,
1792–1814.
Publisher Full Text
Cherel
Y.,
Kernaléguen
L.,
Richard
P.
&
Guinet
C. 2009.
Whisker isotopic signature depicts migration patterns and multi-year intra- and inter-individual foraging strategies in fur seals. Biology Letters 5,
830–832.
PubMed Abstract | PubMed Central Full Text | Publisher Full Text
Cunningham S.A., Alderson S.G., King B.A. & Brandon M.A. 2003. Transport and variability of the Antarctic Circumpolar Current in Drake Passage. Journal of Geophysical Research—Oceans
108, C58084, doi: 10.1029/2001JC001147.
Daneri
G.A.
&
Carlini
A.R. 2002.
Fish prey of southern elephant seals, Mirounga leonina, at King George Island. Polar Biology 25,
739–743.
Daneri
G.A.,
Carlini
A.R.
&
Rodhouse
P.G.K. 2000.
Cephalopod diet of the southern elephant seal, Mirounga leonina, at King George Island, South Shetland Islands. Antarctic Science 12,
16–19.
Publisher Full Text
de Bruyn
P.J.N.,
Bester
M.N.,
Carlini
A.R.
&
Oosthuizen
W.C. 2009.
How to weigh an elephant seal with one finger: a simple three-dimensional photogrammetric application. Aquatic Biology 5,
31–39.
Publisher Full Text
de la Mare
W.K. 1997.
Abrupt mid-twentieth-century decline in Antarctic sea-ice extent from whaling records. Nature 389,
57–60.
Publisher Full Text
Dragon
A.-C.,
Bar-Hen
A.,
Monestiez
P.
&
Guinet
C. 2012.
Horizontal and vertical movements as predictors of foraging success in a marine predator. Marine Ecology Progress Series 447,
243–257.
Publisher Full Text
Ducklow
H.W.,
Baker
K.,
Martinson
D.G.,
Quetin
L.B.,
Ross
R.M.,
Smith
R.C.,
Stammerjohn
S.E.,
Vernet
M.
&
Fraser
W. 2007.
Marine pelagic ecosystems: the West Antarctic Peninsula. Philosophical Transactions of the Royal Society B 362,
67–94.
Publisher Full Text
Fedak
M.,
Lovell
P.,
McConnell
B.
&
Hunter
C. 2002.
Overcoming the constraints of long range radio telemetry from animals: getting more useful data from smaller packages. Integrative and Comparative Biology 42,
3–10.
PubMed Abstract | Publisher Full Text
Forcada
J.,
Trathan
P.N.,
Boveng
P.L.,
Boyd
I.L.,
Burns
J.M.,
Costa
D.P.,
Fedak
M.,
Rogers
T.L.
&
Southwell
C.J. 2012.
Responses of Antarctic pack-ice seals to environmental change and increasing krill fishing. Biological Conservation 149,
40–50.
Publisher Full Text
Freitas
C.,
Lydersen
C.,
Fedak
M.
&
Kovacs
K.M. 2008.
A simple new algorithm to filter marine mammal Argos locations. Marine Mammal Science 24,
315–325.
Publisher Full Text
Gallon
S.,
Bailleul
F.,
Charrassin
J.-B.,
Guinet
C.,
Bost
C.-A.,
Handrich
Y.
&
Hindell
M. 2013.
Identifying foraging events in deep diving southern elephant seals, Mirounga leonina, using acceleration data loggers. Deep-Sea Research Part II 88–89
14–22.
Publisher Full Text
Gil-Delgado
J.A.,
Villaescusa
J.A.,
Diazmacip
M.E.,
Velazquez
D.,
Rico
E.,
Toro
M.,
Quesada
A.
&
Camacho
A. 2013.
Minimum population size estimates demonstrate an increase in southern elephant seals Mirounga leonina on Livingston Island, maritime Antarctica. Polar Biology 36,
607–610.
Publisher Full Text
Heerah
K.,
Andrews-Goff
V.,
Williams
G.,
Sultan
E.,
Hindell
M.,
Patterson
T.
&
Charrassin
J.-B. 2013.
Ecology of Weddell seals during winter: influence of environmental parameters on their foraging behaviour. Deep-Sea Research Part II 88–89
23–33.
Publisher Full Text
Heywood
K.J.,
Naveira Garabato
A.C.,
Stevens
D.P.
&
Muench
R.D.
2004. On the fate of the Antarctic Slope Front and the origin of the Weddell Front. Journal of Geophysical Research—Oceans
109, C06021, doi: 10.1029/2003JC002053.
Publisher Full Text
Hindell
M.A.,
Slip
D.J.
&
Burton
H.R. 1991.
The diving behaviour of adult male and female southern elephant seals, Mirounga leonina (Pinnipedia: Phocidae). Australian Journal of Zoology 39,
595–619.
Publisher Full Text
Hindell
M.A.,
Slip
D.J.,
Burton
H.R.
&
Bryden
M.M. 1992.
Physiological implications of continuous, prolonged, and deep dives of the southern elephant seal (Mirounga leonina). Canadian Journal of Zoology 70,
370–379.
Publisher Full Text
Hofmeyr
G.J.G.,
Kirkman
S.P.,
Pistorius
P.A.
&
Bester
M.N. 2012.
Natal site fidelity by breeding female southern elephant seals in relation to their history of participation in the winter haulout. African Journal of Marine Science 34,
373–382.
Publisher Full Text
Holm-Hansen
O.,
Naganobu
M.,
Kawaguchi
S.,
Kameda
T.,
Krasovski
I.,
Tchernyshkov
P.,
Priddle
J.,
Korb
R.,
Brandon
M.,
Demer
D.,
Hewitt
R.P.,
Kahru
M.
&
Hewes
C.D. 2004.
Factors influencing the distribution, biomass, and productivity of phytoplankton in the Scotia Sea and adjoining waters. Deep-Sea Research Part II 51,
1333–1350.
Publisher Full Text
Hückstädt
L.A.,
Koch
P.L.,
McDonald
B.I.,
Goebel
M.E.,
Crocker
D.E.
&
Costa
D.P. 2012.
Stable isotope analyses reveal individual variability in the trophic ecology of a top marine predator, the southern elephant seal. Oecologia 169,
395–406.
Publisher Full Text
James
B.S.,
McIntyre
T.,
Tosh
C.A.,
Bornemann
H.,
Plötz
J.
&
Bester
M.N. 2012.
Inter-population differences in diving behaviour of adult male southern elephant seals (Mirounga leonina). Polar Biology 35,
1759–1766.
Publisher Full Text
Kahru
M.,
Mitchell
B.G.,
Gille
S.T.,
Hewes
C.D.
&
Holm-Hansen
O.
2007. Eddies enhance biological production in the Weddell–Scotia Confluence of the Southern Ocean. Geophysical Research Letters
34, L14603, doi: 10.1029/2007GL030430.
Kuhn
C.E.,
Johnson
D.S.,
Ream
R.R.
&
Gelatt
T.S. 2009.
Advances in the tracking of marine species: using GPS locations to evaluate satellite track data and a continuous-time movement model. Marine Ecology Progress Series 393,
97–109.
Publisher Full Text
Lewin-Koh
N.J.
&
Bivand
R. 2012. maptools: tools for reading and handling spatial objects. R package version 0.8–20. Accessed on the internet at http://cran.r-project.org/web/packages/maptools/index.html in February 2013.
Lewis
M.,
Campagna
C.
&
Quintana
F. 1996.
Site fidelity and dispersion of southern elephant seals from Patagonia. Marine Mammal Science 12,
138–147.
Publisher Full Text
Massom
R.A.
&
Stammerjohn
S.E 2010.
Antarctic sea ice change and variability—physical and ecological implications. Polar Science 4,
149–186.
Publisher Full Text
McConnell
B.J.,
Chambers
C.
&
Fedak
M.A. 1992.
Foraging ecology of southern elephant seals in relation to bathymetry and productivity of the Southern Ocean. Antarctic Science 4,
393–398.
Publisher Full Text
McIntyre
T.,
Ansorge
I.J.,
Bornemann
H.,
Plötz
J.,
Tosh
C.A.
&
Bester
M.N. 2011.
Elephant seal dive behaviour is influenced by ocean temperature: implications for climate change impacts on an ocean predator. Marine Ecology Progress Series 441,
257–272.
Publisher Full Text
McIntyre
T.,
Bornemann
H.,
Plötz
J.,
Tosh
C.A.
&
Bester
M.N. 2011.
Water column use and forage strategies of female southern elephant seals from Marion Island. Marine Biology 158,
2125–2139.
Publisher Full Text
McIntyre
T.,
Bornemann
H.,
Plötz
J.,
Tosh
C.A.
&
Bester
M.N. 2012.
Deep divers in even deeper seas: habitat use of male southern elephant seals from Marion Island. Antarctic Science 24,
561–570.
Publisher Full Text
McIntyre
T.,
de Bruyn
P.J.N.,
Ansorge
I.J.,
Bester
M.N.,
Bornemann
H.,
Plötz
J.
&
Tosh
C.A. 2010.
A lifetime at depth: vertical distribution of southern elephant seals in the water column. Polar Biology 33,
1037–1048.
Publisher Full Text
McIntyre
T.,
Stansfield
L.J.,
Bornemann
H.,
Plötz
J.
&
Bester
M.N. 2013.
Hydrographic influences on the summer dive behaviour of Weddell seals Leptonychotes weddellii in Atka Bay, Antarctica. Polar Biology 36,
1693–1700.
Publisher Full Text
Meredith
M.P.,
Nicholls
K.W.,
Renfrew
I.A.,
Boehme
L.,
Biuw
M.
&
Fedak
M. 2011.
Seasonal evolution of the upper-ocean adjacent to the South Orkney Islands, Southern Ocean: results from a “lazy biological mooring.”. Deep-Sea Research Part II 58,
1569–1579.
Publisher Full Text
Muelbert
M.M.C.,
de Souza
R.B.,
Lewis
M.N.
&
Hindell
M.A. 2013.
Foraging habitats of southern elephant seals, Mirounga leonina, from the northern Antarctic Peninsula. Deep-Sea Research Part II 88–89
47–60.
Publisher Full Text
Mulvaney
R.,
Abram
N.J.,
Hindmarsh
R.C.A.,
Arrowsmith
C.,
Fleet
L.,
Triest
J.,
Sime
L.C.,
Alemany
O.
&
Foord
S. 2012.
Recent Antarctic Peninsula warming relative to Holocene climate and ice-shelf history. Nature 489,
141–144.
PubMed Abstract | Publisher Full Text
Naveen
R.,
Lynch
H.J.,
Forrest
S.,
Mueller
T.
&
Polito
M. 2012.
First direct, site-wide penguin survey at Deception Island, Antarctica, suggests significant declines in breeding chinstrap penguins. Polar Biology 35,
1879–1888.
Naveira Garabato
A.C.,
Heywood
K.J.
&
Stevens
D.P. 2002.
Modification and pathways of Southern Ocean Deep Waters in the Scotia Sea. Deep-Sea Research Part I 49,
681–705.
Publisher Full Text
Newland
C.,
Field
I.C.,
Cherel
Y.,
Guinet
C.,
Bradshaw
C.J.A.,
McMahon
C.R.
&
Hindell
M.A. 2011.
Diet of juvenile southern elephant seals reappraised by stable isotopes in whiskers. Marine Ecology Progress Series 424,
247–258.
Publisher Full Text
Nicol
S.,
Foster
J.
&
Kawaguchi
S. 2012.
The fishery for Antarctic krill—recent developments. Fish and Fisheries 13,
30–40.
Publisher Full Text
Orsi
A.H.,
Whitworth
T. III
&
Nowlin
W.D.J. Jr. 1995.
On the meridional extent and fronts of the Antarctic Circumpolar Current. Deep-Sea Research Part I 42,
641–673.
Publisher Full Text
Park
Y.-H.,
Charriaud
E.
&
Fieux
M. 1998.
Thermohaline structure of the Antarctic Surface Water/Winter Water in the Indian Sector of the Southern Ocean. Journal of Marine Systems 17,
5–23.
Publisher Full Text
Park
J.,
Oh
I.-S.,
Kim
H.-C.
&
Yoo
S. 2010.
Variability of SeaWiFs chlorophyll-a in the southwest Atlantic sector of the Southern Ocean: strong topographic effects and weak seasonality. Deep-Sea Research Part I 57,
604–620.
Publisher Full Text
Pinheiro
J.C.
&
Bates
D.M. 2004. Mixed-effects models in S and S-Plus. New York: Springer.
Pinheiro
J.,
Bates
D.,
DebRoy
S.
&
Sarkar
D. 2012. nlme: linear and nonlinear mixed effects models. R package version 3.1–105. Accessed on the internet at http://cran.r-project.org/web/packages/nlme/index.html in February 2013.
Pollard
R.T.,
Lucas
M.I.
&
Read
J.F. 2002.
Physical controls on biogeochemical zonation in the Southern Ocean. Deep-Sea Research Part II 49,
3289–3305.
Publisher Full Text
R Core
Team 2012. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing.
Robinson
P.W.,
Simmons
S.E.,
Crocker
D.E.
&
Costa
D.P. 2010.
Measurements of foraging success in a highly pelagic marine predator, the northern elephant seal. Journal of Animal Ecology 79,
1146–1156.
PubMed Abstract | Publisher Full Text
Schlitzer
R. 2002.
Interactive analysis and visualization of geoscience data with Ocean Data View. Computers & Geosciences 28,
1211–1218.
PubMed Abstract | PubMed Central Full Text | Publisher Full Text
Schulte-Hostedde
A.I.,
Zinner
B.,
Millar
J.S.
&
Hickling
G.J. 2005.
Restitution of mass-size residuals: validating body condition indices. Ecology 86,
155–163.
Publisher Full Text
Siniff
D.B.,
Garrott
R.A.,
Rotella
J.J.,
Fraser
W.R.
&
Ainley
D.G. 2008.
Projecting the effects of environmental change on Antarctic seals. Antarctic Science 20,
425–435.
Publisher Full Text
Spreen
G.,
Kaleschke
L.
&
Heygster
G.
2008. Sea ice remote sensing using AMSR-E 89-GHz channels. Journal of Geophysical Research—Oceans
113, C02S03, doi: 10.1029/2005JC003384.
Publisher Full Text
Takahashi
A.,
Matsumoto
K.,
Hunt
G.L.,
Schultz
M.T.,
Kitaysky
A.S.,
Sato
K.,
Iida
K.
&
Watanuki
Y. 2008.
Thick-billed murres use different diving behaviours in mixed and stratified waters. Deep-Sea Research Part II 55,
1837–1845.
Publisher Full Text
Tosh
C.A.,
Bornemann
H.,
Ramdohr
S.,
Schröder
M.,
Martin
T.,
Carlini
A.,
Plötz
J.
&
Bester
M.N. 2009.
Adult male southern elephant seals from King George Island utilize the Weddell Sea. Antarctic Science 21,
113–121.
Publisher Full Text
Trivelpiece
W.Z.,
Hinke
J.T.,
Miller
A.K.,
Reiss
C.S.,
Trivelpiece
S.G.
&
Watters
G.M. 2011.
Variability in krill biomass links harvesting and climate warming to penguin population changes in Antarctica. Proceedings of the National Academy of Sciences of the United States of America 108,
7625–7628.
PubMed Abstract | PubMed Central Full Text | Publisher Full Text
Whitworth
T. III
&
Nowlin
W.D. Jr. 1987.
Water masses and currents of the Southern Ocean at the Greenwich Meridian. Journal of Geophysical Research—Oceans 92,
6462–6476.
Publisher Full Text
Whitworth
T. III,
Nowlin
W.D. Jr.,
Orsi
A.H.,
Locarnini
R.A.
&
Smith
S.G. 1994.
Weddell Sea Shelf Water in the Bransfield Strait and Weddell–Scotia Confluence. Deep-Sea Research Part I 41,
629–641.
Publisher Full Text